Open Colorimeter Introduction
Colorimeters are extremely flexible and easy to use lab instruments. Below are just some examples of how colorimeters are used:
- Water quality testing: there are a wealth of colorimetric assays for measuring water parameters such as water turbidity, chlorine content, pH, water hardness, phosphate content and more
- Monitoring freshwater & saltwater habitats: wildlife and conservation biologists as well as aquarium & aquaponics hobbyists can use colorimeters to monitor levels of ammonia, nitrate, nitrite, phosphate in a habitat
- Soil measurements: colorimetric assays can be used to measure environmental pollutants and contaminants such as lead, oil, diesel and pesticides in soil
- Biochemical assays: Standard methods in labs for measuring protein employs the Bradfords reagent and measurement assay
How colorimeters work
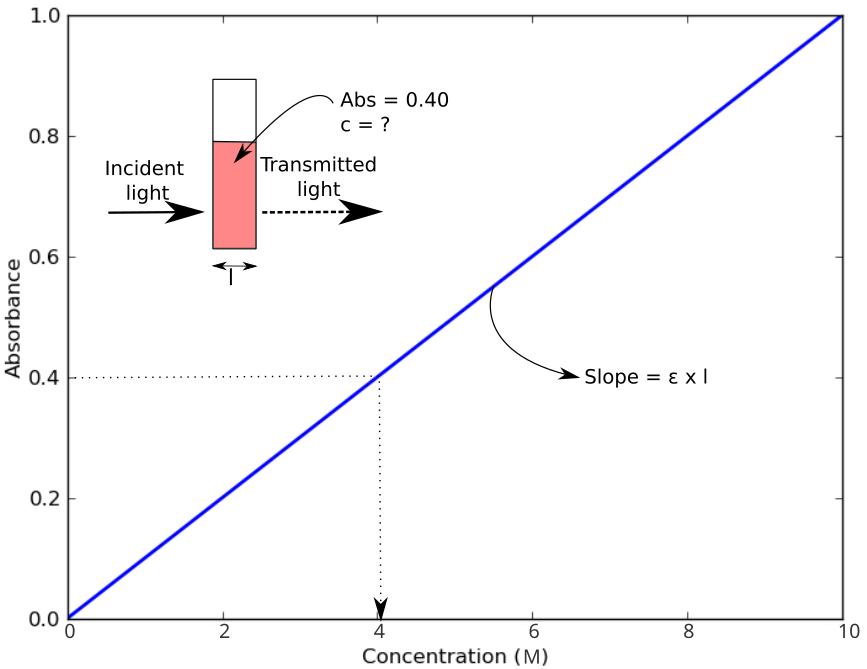
Colorimeters are used to quantify the concentration of an analyte in a sample based on the amount of light of a specific wavelength that is absorbed by the test sample. Absorbance is related to concentration as described by the Beer-Lambert Law. The greater the concentration of a sample, the greater the absorbance value. A light sensor measures the amount of light absorbed by the sample. Comparison of the absorbance measurements to a calibration curve provides the final concentration in the sample.
Absorbance (A) of the sample is determined by comparing the intensity of incident light (I0) to the intensity of light after it has passed through the sample (I) using the following equation:
$$ A = -\mathrm{log}_{10}(I/I_0)$$

Open Colorimeter Product Guide
Documentation site for the Open Colorimeter, Multi-Channel Colorimeter, UV Open Colorimeter & Open Colorimeter Plus
Product Guide Home Page